Chapter 2 Acids, Bases and Salts Class 10 Science Extra Questions

Chapter 2 Acids, Bases and salts Extra Questions and Answer
Chapter Name | Acids, Bases and Salts Extra Questions |
Class | CBSE Class 10 |
Textbook Name | Acids, Bases and Salts Class 10 |
Related Readings |
|
Question 1. Write a balanced chemical equation for the reaction between sodium carbonate and hydrochloric acid indicating the physical state of the reactants and products. [Foreign 2010]
Answer
Na2CO3 (s) + 2HCl (ag) → 2NaCl (aq) + CO2 (g) + H2O (l)
Question 2. During summer season, a milkman usually adds a small amount of baking soda to fresh milk. Give reason. [CBSE Sample Paper 2009]
Answer
A milkman adds a very small amount of baking soda so as to prevent spoilage of milk. It leads to change in the pH which does not allow bacteria and enzymes to act and milk does not become sour due to fermentation.
Question 3. What is the difference between slaked lime and lime water? [CBSE 2010]
Answer
A suspension of Ca(OH)2 in water is called slaked lime. Water containing traces of Ca(OH)2 is called lime water.
Question 4. Which acid is present in sour milk or curd?
Answer
Lactic acid.
Question 5. Why is potassium iodide added into common salt to use it as table salt?
Answer
The iodide present in the salt prevents thyroid disorders.
Question 6. What are the pH values of distilled water and common salt solution? [CBSE 2010]
Answer
Both are neutral and have pH close to 7.
Question 7. A dry pellet of a common base B, when kept in open absorbs moisture and turns sticky. The compound is also a by-product of chloralkali process. Identify B. What type of reaction occurs when B is treated with an acidic oxide? Write a balanced chemical equation for one such solution. [NCERT Exemplar]
Answer
Dry pellets of sodium hydroxide absorb moisture and turn sticky when kept in open which is also a by-product of chloralkali process.
When sodium hydroxide is treated with an acidic oxide it produces salt and water.![]()
Question 8. Which bases are called alkalies? Give an example of an alkali. [CBSE 2009, 2010]
Answer
Soluble bases are called alkalies. For example, sodium hydroxide (NaOH).
Question 9. A knife, which is used to cut a fruit, was immediately dipped into water containing drops of blue litmus solution. If the colour of the solution is changed to red, what inference can be drawn about the nature of the fruit and why? [CBSE 2011]
Answer
Since the colour of the blue litmus has changed to red, this means that the fruit juice is acidic in nature.
Question 10. How do H+ ions exist in water?
Answer
H+ ions in water combine with water (H2O) molecules and exist as H3O+ ion, called hydronium ion.
Question 11. What should be done as remedy if stung by leaves of nettle plant in the wild?
Answer
The area should be rubbed with the leaf of dock plant.
Question 12. What happens when nitric acid is added to egg shell? [NCERT Exemplar]
Answer
Egg shell is made of calcium carbonate. When nitric acid is added to egg shell calcium nitrate, carbon dioxide and water are formed.
CaCO3 + 2HNO3 → Ca(NO3)2 + CO2 + H2O
Question 13. What is the concentration of H+ ion in pure water?
Answer
10-7
Question 14. Which one of these has a higher concentration of H+ ions? 1 M HCl or 1 M CH3COOH. [CBSE 2009]
Answer
1 M HCl has higher concentration of H+ ions.
Question 15. Name an example of olfactory indicators.
Answer
Vanilla.
Question 16. Name the chemical substance present in thick white and yellowish clouds present in the atmosphere of Venus.
Answer
Sulphuric acid.
Question 17. What is acid rain?
Answer
Rainwater having pH less than 5.6, is called acid rain.
Question 18. Name the hardest substance in the body.
Answer
Tooth enamel (Calcium phosphate).
Question 19. The pH of three solutions A, B and C are 4, 9 and 6 respectively. Arrange them in increasing order of acidic strength. [CBSE 2010]
Answer
The increasing order of acidic strength is : B < C < A.
Question 20. Name the chemist who had given the pH scale.
Answer
S.P.L. Sorensen (1909).
Question 21. Name the acid present in tomato.
Answer
Oxalic acid.
Question 22. Acidic and basic solutions in water conduct electricity. Why?
Answer
Because they produce hydrogen and hydroxide ions respectively.
Question 23. What would be the colour of litmus in a solution of sodium carbonate? [CBSE 2009]
Answer
Red litmus will change to blue in sodium carbonate solution.
Question 24. The pH of a sample of vegetable soup was found to be 6.5. How is this soup likely to taste?
Answer
The taste will be slightly sour as it is weakly acidic.
Question 25. Name the chemical substance which is used in the manufacture of soap as well as used as a preservative in pickles.
Answer
Sodium chloride (NaCl).
Question 26. There are two jars A and B containing food materials. Food in jar ‘A’ is pickled with acetic acid while ‘B’ is not. Food of which of jar will stale first? Explain. Name two synthetic indicators which are used to test acids and bases.
Answer
Food in jar ‘B’ will stale first because it will undergo oxidation and will also be attacked by microorganisms.
Synthetic indicators: Phenolphthalein, methyl orange.
Question 27. What is the chemical formula of soda ash?
Answer
Na2CO3
Question 28. Name the substance used for disinfecting drinking water supply.
Answer
Bleaching powder.
Question 29. Name a chemical substance which can be used to detect the presence of moisture in a liquid.
Answer
Anhydrous copper sulphate.
Question 30. What is meant by water of crystallisation?
Answer
Water of crystallisation is the fixed number of water molecules chemically attached to each formula unit of a salt in its crystalline form.
Question 31. Which one is a stronger acid, with pH = 5 or with pH = 2?
Answer
The acid with pH = 2 is a stronger acid.
Question 32. Fresh milk has a pH of 6. When it changes into curd (yogurt), will its pH value increases or decrease? Why?
Answer
Its pH will decrease because curd (yogurt) is sour in taste due to presence of acid in it.
Short Answer Type Questions
Question 1. How would you distinguish between baking powder and washing soda by heating? [NCERT Exemplar]
Answer
Baking soda (NaHCO3) gives carbon dioxide and water vapour on heating at very low temperature. The gas so formed turns lime water milky, which confirms the presence of carbon dioxide gas.
When washing soda (Na2CO3) is heated it does not produce carbon dioxide even at high temperatures, but gives off its water of crystallisation to become anhydrous salt.
Question 2. A sulphate salt of Group 2 element of the Periodic Table is a white, soft substance, which can be moulded into different shapes by making its dough. When this compound is left in the open for some time, it becomes a solid mass and cannot be used for moulding purposes. Identify the sulphate salt and why does it show such a behaviour? Give the reaction involved. [NCERT Exemplar]
Answer
Calcium belongs to group 2. Calcium sulphate is a white soft substance. It is known as Plaster of Paris, which can be moulded into different shapes by making its dough.
When Plaster of Paris is left for some time in the open, it turns into a solid mass because of reaction with moisture present in the atmosphere. The solid mass so formed is known as gypsum and cannot be further used for moulding.
The above said group 2 element is calcium sulpahte.
Question 3. Name the acid present in ant sting and give its chemical formula. Also give the common method to get relief from the discomfort caused by the ant sting. [NCERT Exemplar]
Answer
The acid present in ant sting: Methanoic acid
Chemical Formula of methanoic acid: HCOOH
Method to get relief from the discomfort caused by the ant sting: Rubbing baking soda over the area of ant sting.
Explanation: Rubbing baking soda (a base) over ant sting neutralises the methanoic acid present in the ant sting and gives relief from pain.
Question 4. List two differences between acids and bases on the basis of chemical properties.
Answer
(i) Dilute acids like HCl and H2SO4 evolve H2 gas on reacting with metals like Zn, Mg and Ca, etc. and dilute bases do not evolve hydrogen gas.
(ii) Acids react with oxides of metals while bases react with oxides of non-metals.
Question 5. List four main differences between acids and bases.
Answer
|
Properties |
Acids |
Bases |
|
1. Taste |
Sour |
Bitter |
|
2. Action on litmus paper |
They turn blue litmus paper red |
They turn red litmus paper blue. |
|
3. Action with phenolphthalein |
No action |
They turn phenolphthalein pink. |
|
4. Action with carbonates and bicarbonates |
They decompose carbonates and bicarbonates to liberate carbon dioxide. |
No action |
Question 6. Mention the terms defined by the following sentences:
(a) A soluble base
(b) The insoluble solid formed when two solution are mixed together.
Answer
(a) Alkali
(b) Precipitate.
Question 7. Name the product formed in each case when:
(а) hydrochloric acid reacts with caustic soda.
(b) granulated zinc reacts with caustic soda.
Answer
(a) The product formed is a mixture of sodium chloride and water.
NaOH (ag) + HCl (ag) → NaCl (ag) + H2O
(b) The product formed is a mixture of sodium zincate and hydrogen gas.
Zn (s) + 2NaOH (ag) → Na2ZnO2 (ag) + H2(g)
Question 8. Explain why sodium hydroxide solution cannot be kept in aluminium containers? Write equation for the reaction that may take place for the same.
Answer
Sodium hydroxide solution reacts with aluminium to form sodium metaaluminate and hydrogen is evolved. Therefore, it cannot be kept in a container made of aluminium.![]()
Question 9. How can you obtain the following gases by using dilute acid and one other substance?(а) hydrogen
(b) carbon dioxide.
Answer
(a) Fe + H2SO4(dil.) → FeSO4 + H2(g)
Mg + 2HCl(dil.) → MgCl2 + H2(g)
(b) Na2CO3 + 2HCl(dil.) → 2NaCl + H2O + CO2(g)
NaHCO3 + HCl(dil.) → NaCl + H2O + CO2(g)
Question 10. A solution of HCl is taken in a beaker and an electric circuit with a bulb is set up with the solution in series. What happens to the bulb and why?
Answer
The bulb will start glowing. Glowing of the bulb indicates that there is a flow of electric current through the solution. Electric current is carried through the solution by ions.
Since the cation present in acids is H+, this suggests that acids produce hydrogen ions, H+(ag), in solution, which are responsible for carrying current through the solution.
Question 11. If 280 g of washing soda crystals are left in dry air for some time, a loss of weight of 162 g occurs. How can you account for this?
Answer
Washing soda (Na2CO3. 10H2O) is an efflorescent substance (if exposed to air, it loses most of its water of crystallisation). 280 g of washing soda lose 162 g of its water of crystallisation.
Question 12. A sample of bleaching powder was kept in an air tight container. After a month, it lost some of its chlorine content. How will you account for it?
Answer
Bleaching powder if kept even in an air tight container, will slowly decompose on its own and form calcium chlorate and calcium chloride. The reaction is called auto oxidation. This will result in decrease in its chlorine contents.
![]()
Question 13. A compound which is prepared from gypsum has the property of hardening when mixed with proper quantity of water. Identify the compound. Write chemical equation to prepare the compound. Mention one important use of the compound.
Answer
The compound is Plaster of Paris (CaSO4. ½H2O). It is formed from gypsum (CaSO4 . ½H2O) upon heating to a temperature of 373 K. It changes back to gypsum on adding water. Plaster of Paris is used for setting fractured bones.
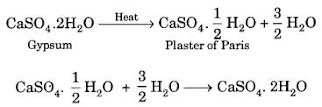
Question 14. A white powder is added while baking breads and cakes to make them soft and fluffy. Write the name of the powder. Name its main ingredients. Explain the function of each ingredient. Write the chemical reaction taking place when the powder is heated during baking. [CBSE 2012, 2013]
Answer
The white powder is known as baking powder. The main ingredients are baking soda (NaHCO3) and tartaric acid (C4H6O6).
Question 16. Explain giving reasons:
(i) Tartaric acid is a component of baking powder used in making cakes.
(ii) Gypsum (CaSO4. 2H2O) is used in the manufacture of cement.
Answer
(i) Role of tartaric acid in baking powder (mixture of tartaric acid and sodium hydrogen carbonate is to neutralise sodium carbonate formed upon heating sodium hydrogen carbonate.
In case it is not done, cake will be better and sodium carbonate will also have injurious side effects.
(ii) The role of gypsum (CaSO4.2H2O) in the manufacture of cement is to slow down the process of setting of cement.
